The Secret Science of Illegal Chemistry


Underneath your feet there is a secret world where science itself is so threatening that it must be kept hidden. This is walk through the science of clandestine chemistry and exposes the hidden chemicals, techniques and knowledge used to produce the illegal drugs consumed by millions around the world that authorities have spent decades trying to prevent amateur cartel bosses discovering.
“You can’t make drugs in your kitchen. It’s an urban myth. if users saw what these chemicals do to instruments in a lab, they wouldn’t put them anywhere near your nose, mouth or stomach. That’s why cooks typical don’t take the drugs they produce”, says the mysterious figure sitting across the table in a haze of cigarette smoke. He continues, “You can tell sloppy chemistry work by the quality of the powder — acid salts tend to be white and crystalline, almost like table salt. If you have been sold drugs that are discoloured, melt away, adsorb moisture from the air, clump into tiny blocks or smell strongly, then it’s crappy lab processes.”
Don’t try to make any of these substances through the information given here. You either end up being raided by the police, injuring yourself and in all likelihood, killing yourself through a laboratory accident or direct poisoning. If you don’t have professional experience in organic chemistry and go about trying to do it in an amateur environment, you really have a death wish. Most labs are found not only due to their fumes and their ordering paper trail, but because of an accident that’s happened. A large degree of cooks are killed in their own labs by processes gone very wrong, typically explosions or poisoning.
Dealing with Ergot alkaloids and the processes involved in synthesising LSD-25 is as about as dangerous and complicated as you can get — for example, unshielded contact with Ergotamine can bring on instant labour in women (even when they’re not pregnant). The next step up is producing nerve gas.
Illegal drug labs are known as “clan labs”, all of which are traditionally run by a “cook”. The chemist community is very tightly knit, and charmingly there is a camaraderie between the police chemists and the clandestine kind as they have a fascination with chemistry in common.
Almost all illegal drugs have been created, re-created and re-synthesised in multiple ways by established credible scientists — they were created originally for specific purposes and mostly only made illegal when they came to be socially abused. Each time a scientist or lab team perfects a new method to make any interesting compound, they generally publish it in one or more scientific journals (e.g. Merck Index, Journal of Forensic Science/JFS, Journal of Organic Chemistry/JOC, Journal of the American Chemical Science/JACS, Journal of Medicinal Chemistry/JMC etc). These can be found in any university library, so the “recipes” to make them are taken directly from these journals and expanded in to work in clandestine labs.
Nowadays most mass industrial illegal drug production in Europe is controlled by criminal gangs centred around 2 places: the first being Holland, and the second being Eastern Europe (the former Baltic states etc). Up until a few years ago Holland had very lax laws on analogues of MDMA (MDA, MDE etc) combined with a liberal tolerance for soft drugs, and is also an ideal spot as a production estuary and for international trafficking connections. The labs themselves are typical mobile and not fixed to a specific location — roaming mobile homes, caravans and barges make ideal facilities.
The influence of Eastern Europe is largely due to the lawless breakdown of the Baltic states, and the influx of highly-educated but unemployed scientists from Russia migrating into the illicit trade by promises of riches in exchange for them doing extremely simple lab procedures. The gangs themselves control the black market in chemical and weapons supplies, so getting hold of controlled precursors by bribing and theft is far easier than setting up lab operations to regress back through generating the precursors via multiple chemical steps.
Chemistry 101: The role of precursors
And all drugs are generally formed from a chemical intermediate, known as a “precursor”. A precursor is the step before the main product — the template material that gets converted and changed into the final compound. All other steps beforehand are focused on creating this precursor. The precursors for illegal drugs are all very well known and watched heavily, meaning that if you try to get your hands on them, you’re guaranteed a police visit. The battle between the illegal labs and the authorities is one where they have to keep “scaling back” to make the chemicals that then make the precursor that then can be converted.
The INCB also keeps a list of the illegal drug precursors that chemical companies should immediately be suspicious of, and the supply and movement of those chemicals is very closely monitored and watched. Most have no real legitimate use other than for producing illicit drugs.
https://www.incb.org/documents/PRECURSORS/RED_LIST/RedList2015_E_14hEd.pdf
Don’t try to order a pill press, Safrole, Ephedrine, Methyl-ecgonine or Phenylacetone.
Au Natural — Extracting from plant material
A sizeable collection of illegal drugs are made simply be extracting them from plants they occur in. Examples are Cannabis, Morphine, Cocaine, Khat, Magic Mushrooms and LSD. Most occur in natural material as “alkaloids” and derive their name from being basic (alkaline). The procedure for extracting an alkaloid from plant material is easy and pretty much the same each time. They are prepared into powders by reacting them with strong acid to form their solid acid salt for storage and eventual ingestion. If hydrochloric acid is used, the drug becomes a hydrochloride; if sulphuric acid is used, it becomes a sulphate, and if nitrate is used (very rarely), it becomes a nitrate. The cocaine you know is actually called cocaine hydrochloride, and it’s free-base (i.e. pure form taken out of the plant as a white solid) is called “crack”.
To extract an alkaloid, plant material is added to an organic solvent such as benzene, ethanol or petroleum ether, where it moves/transfers from the leaves etc into the solvent. The liquid is then drained off to be played with, and the plant material thrown away. Next, either a dilute base or acid is added (depending on which compound is being extracted), which causes the extracted materials to form as a precipitate (a goo-like substance) floating on top of, and around inside the liquid. The solvent is then boiled off (often speeded up with the use of a vacuum) to leave just the goo. Various stages of purification of the goo are repeated to get the maximum quality of the starting material.
Poppers — Amyl/butyl/isobutyl nitrite
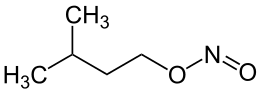
Poppers are a class of chemicals called alkyl nitrites (structure R-ONO) and alkyl esters of nitrous acid. Organic nitrites are generally prepared from alcohols and sodium nitrite in sulfuric acid solution. The illicit creation of semi-legal derivatives is incredibly simple and rather pointless as you can buy the substance legally. What is interesting though is the synthesis of the higher-class nitrites such as Pentyl and Hexyl versions.
Synthesis is a 1-step process — add acidic iso-propyl alcohol (IPA) to sodium nitrite dissolved in water, and decant/dry the result. For pentyl, use Pentan-1-ol, and so on.
GHB — Gamma-Hydroxybutyric acid (4-Hydroxybutanoic acid)
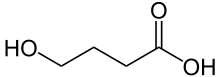
Interestingly, GHB is a naturally-occurring substance that is found in the central nervous system, first being synthesised in 1874. It is extremely easy to produce.
The simplest way to produce GHB is by the hydrolysis (e.g. through the addition of sodium hydroxide) of the corresponding lactone (a cyclic intramolecular ester, e.g. Gamma Butrylactone) to the desired hydroxy acid. Ester hydrolysis can be done in two ways: An acid catalyzed reaction or a base catalyzed reaction. The base catalyzed reaction is the common choice, because the reaction is not reversible like the acid catalyzed one and therefore we will get higher yields, and the chemist will get the sodium salt of GHB, as the free acid is not stable, and will immediately cyclize into gamma-butyrolactone again.
Methcathinone/Khat — 2-(methylamino)-1-phenyl-propan-1-one Hydrochloride
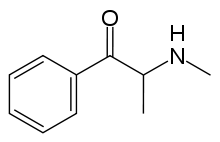
Methcathinone is very similar in structure to ethcathinone, a rare cathinone analogue, and cathinone, a stimulant alkaloid occurring in the shrub Catha edulis (Khat). It has a single chiral carbon atom, thus yielding enantiomeric + and — forms.
Methcathinone is most commonly made by the oxidation of ephedrine or pseudoephedrine, which is the opposite process to the manufacture of amphetamine (reduction of ephedrine). Potassium permanganate (KMnO4) is most commonly used as the oxidant but also considered undesirable because of the low yields and its high toxicity. A method that yields more is oxidizing ephedrine with sodium hypochlorite in glacial acetic acid. As free base is very unstable; it easily loses its ketone status and converts back into an alcohol. Alternative methods include the bromination of propiophenone and addition of Phtalimido-postasium to give phtalimidopropiophenone, which is then reacted with Methylamine for processing into the hydrochloride salt.
Cannabis — 3,4-trans-tetrahydrocannabinol
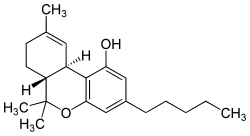
Cannabis (Hemp) is actually a genus rather than a single plant, with 3 separate putative and dioecious species — Cannabis sativa, Cannabis indica and Cannabis ruderalis. Cannabis plants produce a unique family of over 600 terpeno-phenolic compounds called cannabinoids, and the two that are usually produced in greatest abundance are cannabidiol (CBD) and/or delta9-tetrahydrocannabinol (THC), but only THC is psychoactive. As is the case with nicotine and caffeine, the role of THC in Cannabis, it seems, is to protect the plant from herbivores, UV radiation or pathogens. Cross-breeding between species and specific genetic heirlooms of the plant produces the variety of potency. Generally speaking “Skunk” is cultivated from the tall and spindly Sativa, and hashish is created from heating and compressing the resin produced around the female flowers of the smaller, denser indica variety.
Professional labs use hydroponic growing systems (water-based nutrition without soil) and complex fluorescent lighting systems that provide a seed-harvest time of around 12–20 weeks for unfertilised female plants (Sensimillia), depending on the individual species and strain.
Obviously, producing synthetic Cannabinoids is far more expensive, complex and time-consuming than extracting them naturally from the plant itself (THC occurs mainly as its carboxylic acid — THC-COOH). Biosynthesis in the plant is understood to go as follows: the enzymatic condensation of geranyl pyrophosphate and olivetolic acid gives cannabigerolic acid which is cyclized by the enzyme THC acid synthase to give THC-COOH. Heating decarboxylates the acid to THC.
The syntheses of THC-A, THC-B, or THC-C are all based on the condensation of olivetol (5-(n-amyl)-resorcinol) with a second component which forms the left-hand and center ring of the THC molecule. THC-A or THC-B are obtained from olivetol and one of the following; citral, verbenol, or p-2,8-menthadien-2-ol. THC-C is obtained from olivetol and (-)-2-carbethoxy-5-methylcyclohexane. The variants in each of the three series are obtained by replacing olivetol with a 5-(alkyl)-resorcinol in which the alkyl group is altered from n-amyl to one of a number of different additives.
Heroin — Diacetyl-Morphine Hydrochloride
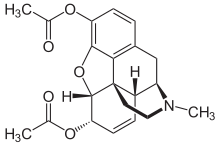
Opium is the name for the milky latex sap/goo produced in the immature seed pods of the white/red flowering poppy Papaver Somniferum. The Phenanthrenes found in the highest concentrations in this sap are Morphine, Codeine and Thebaine. Heroin is the semi-synthetic 3, 6-diacetyl ester of morphine ((5α,6α)-7,8-didehydro-4,5-epoxy-17-methylmorphinan-3,6-diol), and 93% of all opium refinement originates in Afghanistan with 1 acre yielding 25–30kg. Synthetic derivatives of compounds with a morphine-related structure are known as “opioids” or “opiates”.
Production is relatively easy. The collected opium is dissolved in water and made alkaline with lime, removing unwanted impurities. The remaining water filtrate is then acidified with ammonium chloride which generates morphine as a floating white precipitate that can be collected and dried. The resulting substance is then heated with acetic anhydride (acetylation), cooled and diluted in water, and finalised made basic with sodium carbonate, producing raw free-base heroin that can be smoked. The hydrochloride salt of the drug is then created in a standard way with hydrochloric acid in ether, and the precipitate is extracted and retrieved as white solid.
But if you really want to go to town, you create synthetic opiates that are thousands of times more potent than morphine. Welcome to the Fentanyl family — your customers probably won’t survive it. The first kind is 80 times more potent (N-(1-(2-phenylethyl)-4-piperidinyl)-N-phenyl-propanamide and created in 4 steps starting from 4-piperidinone hydrochloride. The ultimate in opiates is Carfentanil, which is used to bring down large mammals (elephants, rhinos etc) as it is 10,000 times more potent than morphine. The Russian government pumped it into the infamous theatre hostage-seize, killing most of the hostages as well as the kidnappers. There are 2 patented syntheses, both which are extremely complex.
Cocaine/Crack — Benzoylmethyl ecgonine Hydrochloride
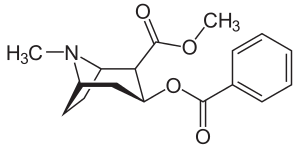
Cocaine is a crystalline tropane alkaloid that is obtained from the leaves of the coca plant Erythroxylon coca. It is a naturally occurring chemical found in certain varieties of plants of the genus Erythroxylum. There are over 200 distinct species of Erythroxylum, of which only two, Erythroxylum coca and Erythroxylum novogranatense, contain significant amounts of cocaine. In South America, two varieties within each of these two species are cultivated; these are Erythroxylum coca var. coca (ECVC), Erythroxylum coca var. ipadu (EM), Erythroxylum novogranatense var. novogranatense (ENVN), and Erythroxylum novogranatense var. truxillense (ENVT). The CIA lists the varities as Huanuco coca, grown in Bolivia and Peru, Amazonian coca, grown in the Amazon River basin, and Colombian coca, grown primarily in Colombia.
Columbia leads the world in its production with 75% market share. Production of illicit natural cocaine involves three steps: 1. Extraction of crude coca paste from the coca leaf; 2. Purification of coca paste to coke base, and 3. Conversion of coke base to cocaine hydrochloride.
Cocaine sulfate is produced by macerating coca leaves along with water that has been acidulated with sulfuric acid, or an aromatic-based solvent, like kerosene or benzene. This is often accomplished by placing the ingredients into a vat and stomping on them, in a manner similar to the traditional method for crushing grapes. After the maceration is completed, the water is evaporated to yield a pasty mass of impure cocaine sulfate. The sulfate salt itself is an intermediate step to producing cocaine hydrochloride.
The low-grade paste is then washed in Kerosene, where the subsequent crystals are dissolved in methanol. Reacting the now free base cocaine with hydrochloric acid gives the pure white hydrochloric acid salt that is snorted, but cannot be smoked as it is destroyed at comparatively low temperatures. To make crack, the hydrochloride salt is regressed back to its free base with sodium hydroxide and/or ammonia, and then precipitated in water.
The basic formula for synthetic cocaine documented by Willstatter starts by purchasing or making tropinone from succinaldehyde, converting it into 2-carbomethoxytropinone (also known as methyl-tropan-3-one-2-carboxylate), reducing this to ecgonine, and changing that to cocaine.
Chemical manipulation of the cocaine molecule to produce higher-performance analogues is focused around strengthening its structure, as it is metabolised too quickly in the human body to become addictive. Certain modifications of natural cocaine can result in products having substantially greater potencies than cocaine. The compounds 2-carbomethoxy-3-(4-fluorophenyl)tropane and 2-carbomethoxy-3-phenylnortropane are both some 60 times more potent than cocaine (Clarke et al. 1973).
Speed/Crystal Meth/Ice — Amphetamine Sulphate / Methamphetamine Hydrochloride
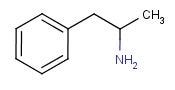
Amphetamine (phenylisopropylamine) was first synthesized in 1887 by Lazar Edeleanu at the University of Berlin and it is one of a series of compounds related to the plant derivative Ephedrine, the main component of slimming pills. The related compound methamphetamine was first synthesized from ephedrine in Japan in 1918 by chemist Akira Ogata via reduction of ephedrine using red phosphorus and iodine. Amphetamines are chiral compounds and are fomed as a racemic mixture that can be divided into its optical antipodes that occur in equal amounts: levo- (l-) and dextro (d-).
The quickest method for synthesis of amphetamine sulphate is reductive amination of Phenyl-2-Propanone (P2P, or Phenylacetone), which is a schedule substance by itself and has little other laboratory use other than for producing amphetamines. As the same process is generally used for producing the more potent Methyl analogue, the lower sulphate is rarely produced.
Because of the schedule nature of Phenylacetone/P2P, most illicit production procedures are focused around finding new and interesting ways to take a step back and produce it in the lab instead of trying to procure it from a supply house.
The reductive amination of phenylacetone (ketone) with methylamine/N-Methyl formamide is known as the Leukardt-Wallach reaction. The reaction requires a catalyst that acts as a reducing agent, such as mercury-aluminum amalgam or platinum dioxide, also known as Adams’ catalyst. Other less common methods use other means of hydrogenation (using an industrial “bomb”), such as hydrogen gas in the presence of a catalyst.
Most methods of illicit production involve hydrogenation of the hydroxyl group on ephedrine or pseudoephedrine (stuffy nose medication). The most common method for small-scale methamphetamine labs is primarily called the “Red, White, and Blue Process“, which involves red phosphorus, pseudoephedrine or ephedrine (white), and blue iodine, from which hydroiodic acid is formed. This method of directly reducing ephedrine, pseudoephedrine, or phenylpropanolamine to meth or benzedrine uses hydrazine hydrate as the reducing agent and is known as the Wolff-Kishner reaction.
An increasingly common method uses the process of Birch reduction, in which metallic lithium, commonly extracted from non-rechargeable lithium batteries is substituted for metallic sodium, to circumvent the difficulty of procuring metallic sodium. Anhydrous ammonia and lithium or sodium may be surpassing hydroiodic acid (catalytic hydrogenation) as the most common method of manufacturing.
An alternative is the Ritter Reaction, is a reaction whereby amides are made by adding an alkene to a mixture of a nitrile in sulfuric acid. After the amide is made, it is then boiled in hydrochloric acid solution to give the corresponding amine. Consequently, amphetamines can be synthesised from Allyl Benzene.
Crystal Meth or “Ice” is methamphetamine produced in a way that it resembles a translucent block of material that can be smoked rather than a powder. The most common method is a form of purification by dissolving existing product into an organic solvent like iso-propyl alcohol, and the desiccation and vacuum evaporation to produce large clear crystals.
Ecstasy (MDMA, MDA, MDE) — 3,4-methylenedioxy-N-methylamphetamine
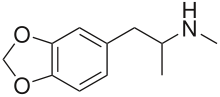
Ecstasy is generally assumed to refer to MDMA, but also contains the analogues MDA and/or MDE. These psychoactive phenethylamine cousins are derived from ampehtamines and considered the original designer drugs. The German company Merck filed the patent for MDMA in 1912, but it became popularised by the work of Alexander Shulgin in his groundbreaking work PIKHAL.
Safrole, a colorless or slightly yellow oil allyl benzene oil, extracted from the root-bark or the fruit of sassafras plants (primarily Ocotea cymbarum, 70% yield) is the primary precursor for almost all manufacture of MDMA. There are numerous synthetic methods available in the literature to convert safrole into MDMA via different intermediates. One common route is via the MDP2P (3,4-methylenedioxyphenyl-2-propanone, also known as piperonyl acetone) intermediate, reflecting a similar process to that of methamphetamine using the P2P precursor. The basic scheme is the oxidation of safrole (1-allyl-3,4-methylenedioxybenzene) into a ketone, which is then condensed with methylamine and reduced to the final MDMA product.
This intermediate can be produced in at least two different ways. One method is to isomerize safrole in the presence of a strong base to isosafrole and then oxidize isosafrole to MDP2P. Another, reportedly better method, is to make use of the Wacker process to oxidize safrole directly to the MDP2P (3,4-methylenedioxy phenyl-2-propanone) ketone intermediate. This can be done with a palladium catalyst. Once the MDP2P intermediate has been produced it is then consumed via a reductive amination to form MDMA as the product.
Other psychedelic amphetamines (Mescaline, 2-CB, DOM etc)
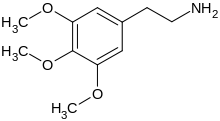
Other “designer” phenethylamines and phenalkylamines include DOB, MMDA, TMA, DMA, PMA, DMHP and DOET.
The fundamental principle governing the production of new phenethylamine analogues is taking compounds found in natural essential oils that can act as amphetamine precursors (i.e. oils that have a smell or “essence”) and reacting them with ammonia to give them an NH3 group. There are some ten essential oils that have a three carbon chain, and each lacks only a molecule of ammonia to become an amphetamine. These essential oils, or “almost” amphetamines, can serve as an index for the corresponding real amphetamine counterparts. These oils include Anethole, Safrole, Apiole, Elemicin, Eugenol, Anise, Nutmeg and many more.
These substitutions follow a few main patterns:
4-Methoxy (Estragole)
3.4 Dimethoxy (Methyleugenol)
3,4 Methylenedixoy (Safrole)
3-methoxy-4,5-methylenedioxy (Myristicin)
2-methoxy-3,4-methylenedioxy (Croweacin)
3,4,5-trimethoxy (Elemicin)
2,4,5-trimethoxy (Asarone)
2,5-dimethoxy-3,4-methylenedioxy (Apiole)
2,3-dimethoxy-4,5-methylenedioxy (Dill Apiole)
Tetramethoxy (Parsley)
Mescaline is a psychedelic alkaloid that occurs naturally in the peyote cactus (Lophophora williamsii), the San Pedro cactus (Echinopsis pachanoi) and the Peruvian Torch cactus (Echinopsis peruviana), and in a number of other members of the Cactaceae. It is also found in small amounts in certain members of the Fabaceae (bean family), including Acacia berlandieri. Mescaline was first isolated and identified in 1897 by the German Arthur Heffter and first synthesized in 1919 by Ernst Späth. There are multiple syntheses, but the primary involves reacting beta-nitro-3,4,5-trimethoxystyrene with sodium tartrate and the converting to the hydrochloride salt.
Indole Alkylamines (Psilocybin, DMT, DET etc)
Over 500 naturally occurring indole alkaloids were known by 1972 and accounted for nearly one fourth of all alkaloids known at that time. By 1980, the number of known indole alkaloids had risen to approximately 1200. The most common indole derivatives are the tryptamine family, of which many brain neurotransmitters are similar in structure to, e.g. Serotonin.
Most tryptamines are found in nature in plants and funghi, so drug-taking itself is either a case of eating/smoking the raw materials, or extracting them with solvents.
DMT was first extracted from the roots of Mimosa hostilis in 1946 by Brazilian ethnobotanist and chemist Gonçalves de Lima who named it Nigerine, and was first synthesized by British chemist Richard Manske in 1931. It is often synthesized by the Speeter-Anthony synthesis from indole using oxalyl chloride, dimethylamine, and lithium aluminium hydride as reagents, and is usually used in its base form, but it is more stable as a salt, e.g. as a fumarate.
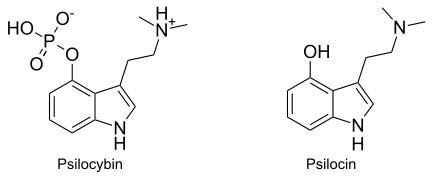
Though DET is a synthetic compound with no known natural sources it has been used with mycelium of Psilocybe cubensis to produce the synthetic chemicals 4-PO-DET and 4-HO-DET, as opposed the naturally occurring 4-PO-DMT (Psilocybin) and 4-HO-DMT (Psilocin). Isolation of the alkaloids resulted in 3.3% 4-HO-DET and 0.01–0.8% 4-PO-DET. It can also be created via the Speeter-Anthony synthesis by substituting dimethylamine with diethylamine.
Psilocybin is the psychoactive compound found in “magic mushrooms” (e.g. Psilocybe azurescens, Psilocybe zapotecorum) and is classed as a prodrug that is converted into the pharmacologically active compound psilocin in the body by dephosphorylation. It is for this reason that the preparation of mushrooms is prohibited, as once they are ingested, psilocin produces the hallucinations.
PCP — Phencyclidine/phenylcyclohexylpiperidine
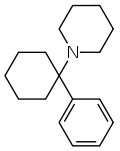
In chemical structure, PCP is an arylcyclohexylamine derivative, and, in pharmacology, it is a member of the family of dissociative anesthetics. It is commonly known as the source of stories concerning people throwing themselves out of windows believing they can fly (not LSD, as widely believed) and that they are invincible.
There are 2 main methods for producing Phencylidine. The first starts with the reaction of cyclohexanone with anhydrous ethylamine, which is then distilled to the intermediate N-cyclohexylidenethylamine. 1- phenylcyclohexylethylamine is obtained by the addition of phenyllithium. The second is to azeotropically and vacuum distil a mixture of piperidine, cyclohexylamine and benzene to obtain cyclohexenyl-piperidine, which is then reacted with p-toluenesulfonic acid monohydrate and PhMgBr for a final decomposition by ammonium hydroxide.
Ketamine — 2-(2-chlorophenyl)-2-methylamino-cyclohexan-1-one Hydrochloride
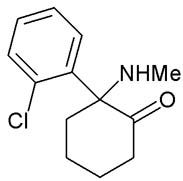
Ketamine is a semi-legal veterinary drug related to PCP and developed by Parke-Davis in 1962, and so generally tends to be stolen and sold on the black market rather than produced illicitly by corrupt Asian pharmaceutical companies. Like amphetamine, it is a chiral and racemic compound with the more active enantiomer, S-ketamine, being the most potent isomer.
The most common synthesis starts with the reaction of cyclopentyl Grignard and o-chlorobenzonitrile to give o-chlorophenyl-cyclopentyl ketone, followed by alpha bromination of the ketone, and then reaction with methylamine to form an alpha-hydroxy imine (1-Hydroxycyclopentyl-(o-chlorophenyl)-ketone-N-methylimine). Heating this imine results in Ketamine via a novel alpha-hydroxyimine rearrangement. Overall yields are around ~60%.
LSD — Lysergic acid diethylamide
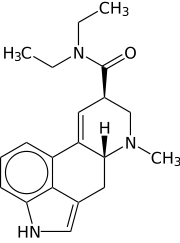
LSD is without a doubt the hardest illegal drug to make as it involves very sophisticated equipment and highly potent compounds that are very unstable. It is also the most powerful hallucinogen in the world, only needing millionths of a gram as a dosage. It is synthesized from lysergic acid derived from ergot, a grain fungus that typically grows on rye and was first synthesized by Swiss chemist Albert Hofmann in 1938. The short form LSD comes from its early codename LSD-25, which is an abbreviation for the German “Lysergsäure-diethylamid” followed by a sequential number.
LSD is an ergoline derivative and all synthesis revolves around the processing of lysergic acid. It is commonly produced from reacting diethylamine with an activated form of lysergic acid. Activating reagents include phosphoryl chloride and peptide coupling reagents. Lysergic acid is made by alkaline hydrolysis of lysergamides like ergotamine, a substance derived from the ergot fungus on rye, or, theoretically, from ergine (lysergic acid amide, LSA), a compound that is found in morning glory (Ipomoea tricolor) and hawaiian baby woodrose (Argyreia nervosa) seeds. LSD is a chiral compound with two stereocenters at the carbon atoms C-5 and C-8, so that theoretically four different optical isomers of LSD could exist.
Beginning with ergotamine tartrate, for example, one can manufacture roughly one kilogram of pure, crystalline LSD from five kilograms of the ergotamine salt. Five kilograms of LSD — 25 kilograms of ergotamine tartrate — could provide 100 million doses. Cooking LSD is time consuming; it takes from 2 to 3 days to produce 1 to 4 ounces of crystal. Consequently, LSD usually is not produced in large quantities, but rather in a series of small batches.
Celebrity Cooks
ALEXANDER SHULGIN
Grandfather of all the designer drugs we know today is the genius Alexander Shulgin, author of “PIKHAL” and “TIKHAL”, which are found in every clan lab without exception. Shulgin is a gentle pensioner who is renowned as the world’s authority on psychedelic drugs, having created over 500 variations of phenyethylamine, tryptamine and quinoline-based compounds.
STRIKE & RHODIUM
The next is “Strike” (Hobart Huson), the creator of a website called “The Hive” where i was a regular poster, and the author of the “Total Synthesis” series of books on MDMA manufacture also found in labs without exception. “Strike” also had a counterpart in the form of a Scandinavian genius chemist known colloquially as “Rhodium”.
UNCLE FESTER
pseudonym for an author who has written a number of underground books (most that were published by the now-defunct Loompanics Unlimited) on many controversial topics such as amphetamine manufacture, poisoning methods and the production of nerve gases.